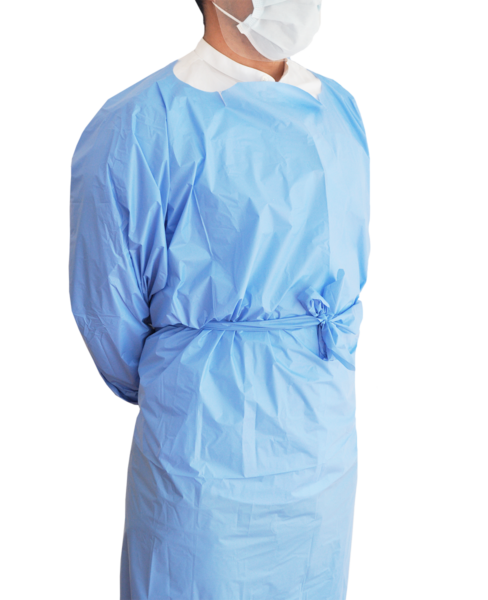
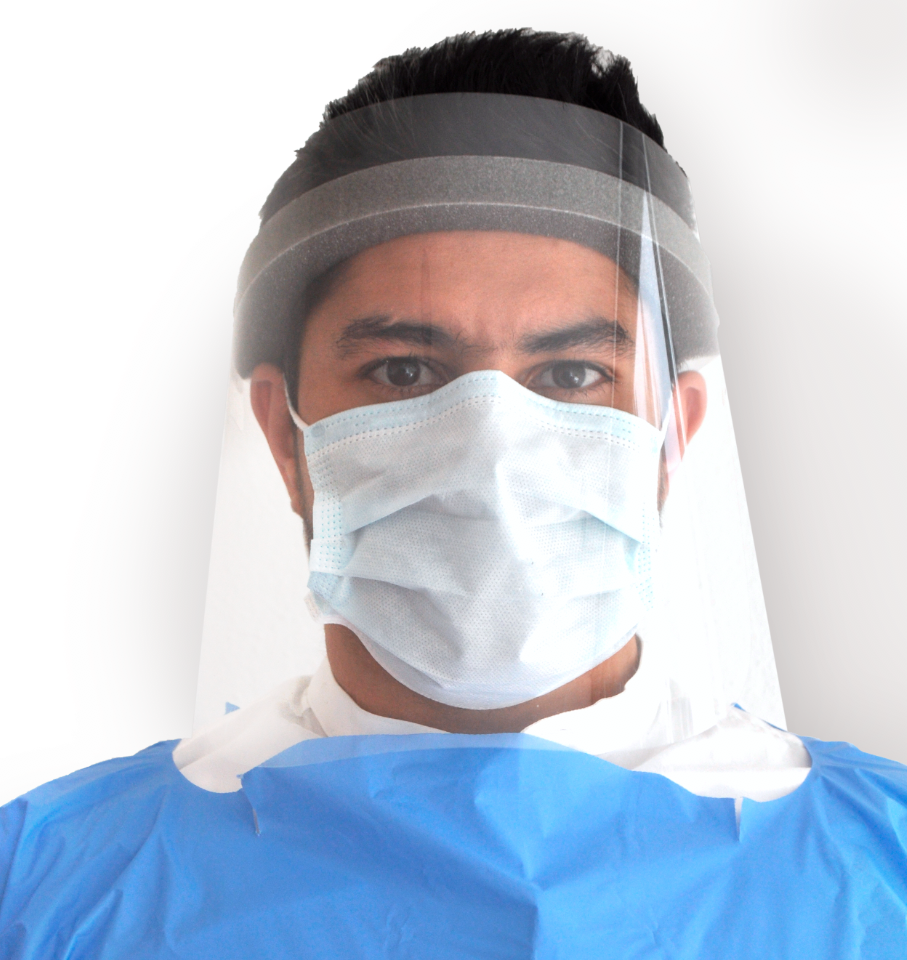
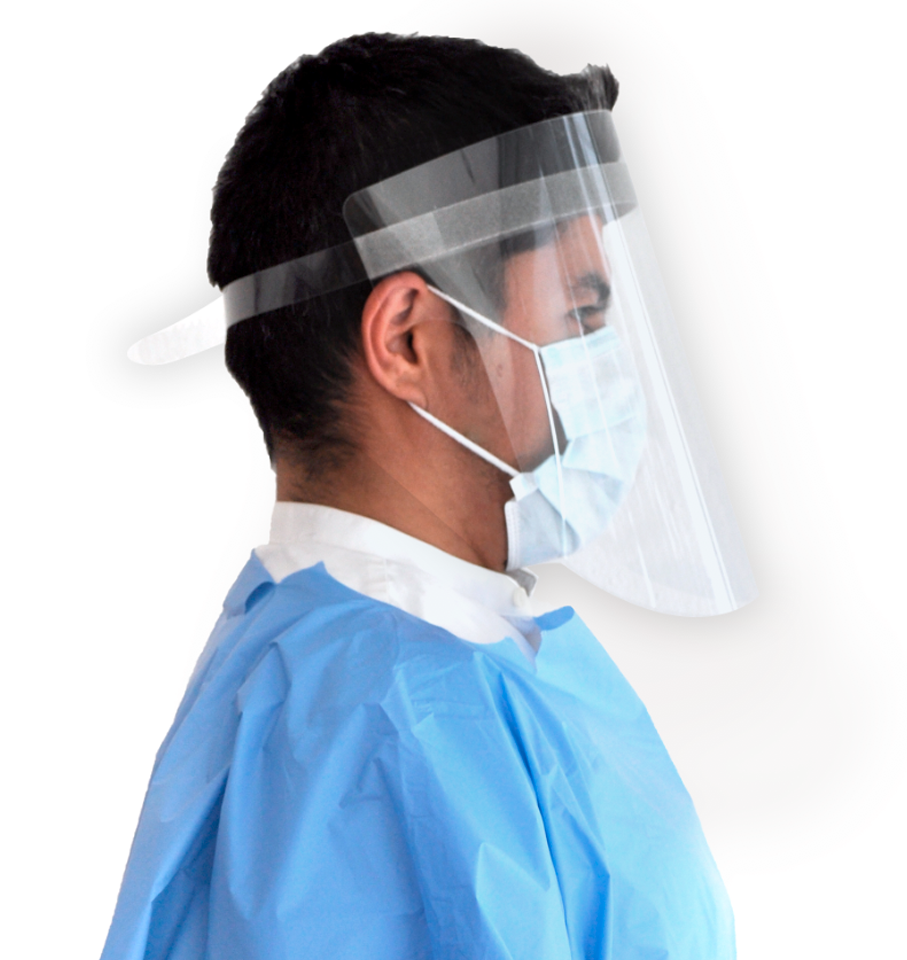
Reliable Protection
Our experience in creating specialty die cut solutions for the automobile market has helped facilitate the manufacturing of reliable cost-effective medical equipment.
With our state-of-the-art production facility and exceptional processing standards, we guarantee our customers compliance with international standards while responding quickly to both low and high-volume orders.